







Example: The $\alpha $ - D - galactose
First step: The pyran cycle is oriented with oxygen behind and right. The carbons 1,2,3,4,5 are numbered from the right in a clockwise direction.

Second step: We write the Fischer → linear structure ($ -CHO $ in position 1 up!):
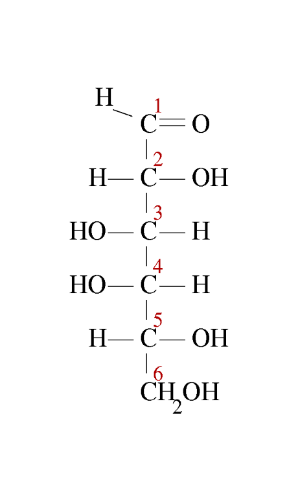
Third step: The $ -CH_2OH $ group, position 6, is attached in position 5 upwards:
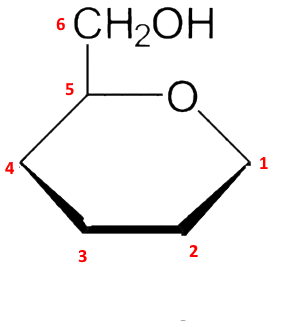
Fourth step: The new $ -OH $ group in position 1, formed by the cyclization, is oriented downwards since one wishes to obtain $\alpha $ the form (for $\beta $ it would be orientated upwards):
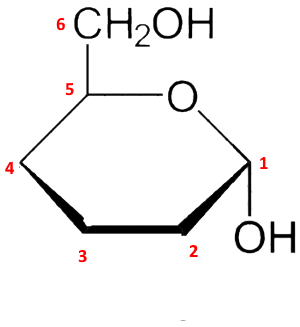
Fifth step: The $ -OH $ groups in the remaining 2,3 and 4 positions are oriented downward or upward depending on whether they are to the right or left in the linear form:
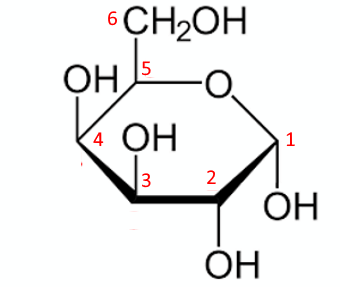
That's it!
Example: The $\beta $ - D - fructose
First step: The furan ring is oriented with oxygen behind and in the center. The carbons 2,3,4,5 are numbered from the right in a clockwise direction.
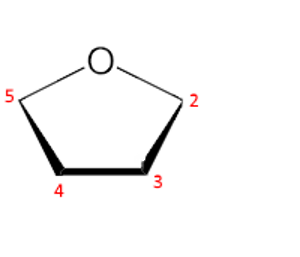
Second step: We write the Fischer → linear structure ($ -CO $ in position 2 up!):
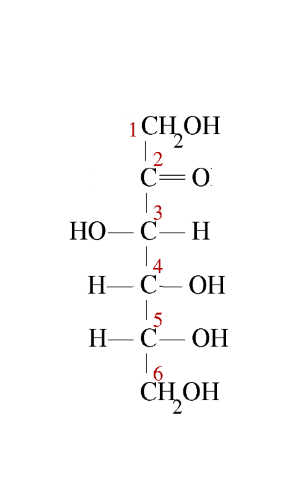
Third step: The $ -CH_2OH $ group, position 6, is attached in position 5 upwards:
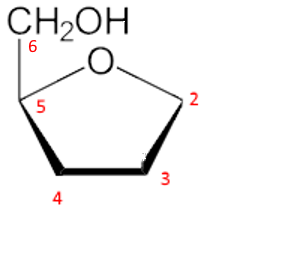
Fourth step: The new $ -OH $ group in position 2, formed by the cyclization, is oriented upward since we want to have the form $\beta $ (for $\alpha $ we would orient down). We don't forget the $CH_2OH$ in position 1.
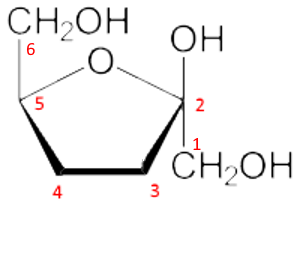
Fifth step: The $ -OH $ groups in the remaining 3 and 4 positions are oriented downward or upward depending on whether they are to the right or left in the linear form:
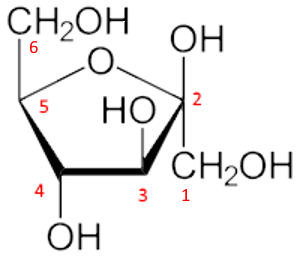
And there you go!