







The cyclization of the two enantiomers..
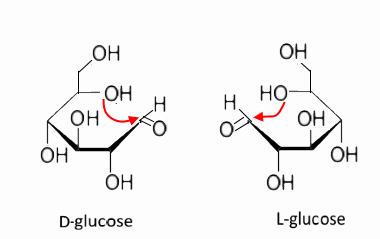
leads to the following two forms: $ \alpha $ ($ OH $ in position 1 down, $ CH_2OH $ in position 6 up!)
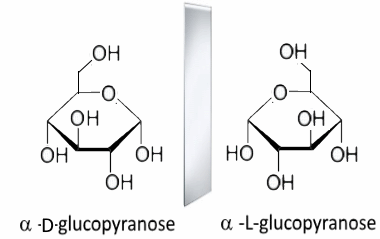
Notice that they are images of each other in a mirror placed vertically between them! By turning the L enantiomer around a horizontal axis perpendicular to our screen, this enantiomer becomes:
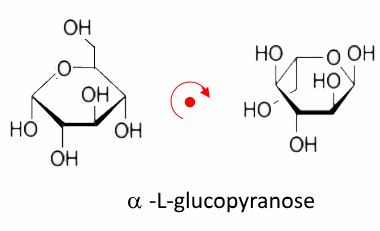
By comparing the D and L forms now, we see that they are also images of each other in a horizontal mirror:
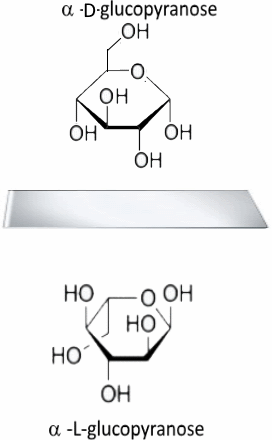
Conclusion:
Starting from the cyclic form D, we obtain the cyclic form L - or by drawing the image in a vertical mirror. Then $ OH $ in position 1 remains pointing downward, $ CH_2OH $ in position 6 up! - or by drawing the image in a horizontal mirror. Then the cycle remains in place ($ -O- $ on the top right!), but all the attached groups change!
In fact, all our previous laborious work comes down to the simple fact that enantiomers are always images of each other in a mirror!