





- Fish and amphibians eliminate excess nitrogen usually in the form of ammonia $NH_3$ which is soluble in water - Reptiles and birds excrete the excess nitrogen in the form of uric acid
 -
- Mammals (including humans) excrete the excess nitrogen as urea
The great Dutch scientist Hermanno Boerhaave isolated for the first time urea from urine
 Hermanno Boerhave 1668-1738
"Simplex sigillum veri; Simplicity is the sign of truth"
Hermanno Boerhave 1668-1738
"Simplex sigillum veri; Simplicity is the sign of truth"

Urea is - a white crystalline solid - non-toxic - very soluble in water
 Friedrich Wöhler 1800-1882
Friedrich Wöhler 1800-1882
In 1828, Friedrich Wöhler synthesized urea from ammonium cyanate: $NH_4^+NCO^- \rightarrow H_2NCONH_2$ The synthesis of this organic substance by an inorganic reactant showed that it is no need of "vis vitalis - vital force" to the mysterious essence of life!
- Basic character The electron pair on the nitrogen atom is rendered less active by the effect of the neighboring carboxyl group. However urea may react as a base eg with nitric acid: $H_2NCONH_2$ $+$ $HNO_3$ $\rightarrow$ $ H_2NCONH_3^+NO_3^-$ (formation of urea nitrate) - Reaction with nitrous acid As in the case of amines, nitrous acid is able to replace the amino groups with hydroxyl groups and so form carbonic acid easily decomposed to water and carbon dioxide $H_2NCONH_2$ $+$ $2HNO_2$ $\rightarrow$ $ H_2CO_3+2N_2$ $\rightarrow$ $ CO_2$ $+$ $H_2O$ $+$ $2N_2$ This reaction can be used to destroy the toxic nitrous acid! - Polycondensation with formaldehyde (methanal)
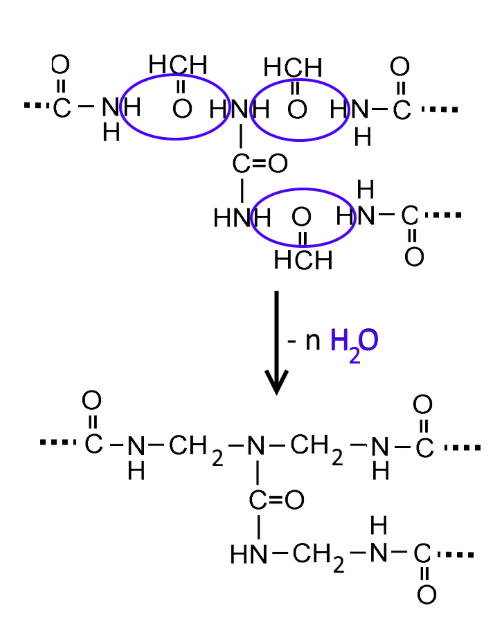
There is formation of a three-dimensional polymer called aminoplast..
Video due to William Escudier
- Aminoplasts (hard formalin-urea resins)

- Plywood and laminate (wood bound by formaldehyde-urea resin)

- Fertilizers

- Skin moisturizers (cosmetics)
