





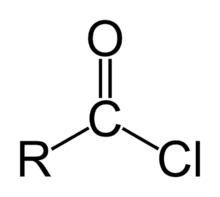
 Acid chloride
Acid chloride
-In contrast to carboxylic acids, they can not form hydrogen bonds between them. Therefore they are more volatile than the corresponding carboxylic acids. - In contact with water, they release $HCl$, hence their lacrymatory effect
Extract of a video due to Franklychemistry
Extract of a video due to Franklychemistry
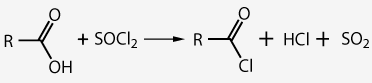
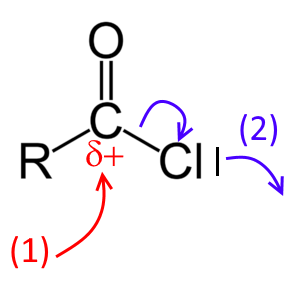
The structure of the functional group of acyl chlorides explains their chemical reactivity:
- (1) The $ C$ atom is an electrophilic center ($ \delta + $ )
These substances therefore undergo nucleophilic attack, for example by $H_2O^{\delta -}$, $RO^{\delta-}H$, $H_3N^{\delta -}$ or $RH_2N^{\delta -}$:
Reaction with water: $RCOCl + H_2O$ $\rightarrow$ $ RCOOH+ HCl$ Product: The corresponding carboxylic acid ! Réaction avec un alcool: $RCOCl + R^{'}OH$ $\rightarrow$ $ RCOOR^{'}+ HCl$ Product: The corresponding ester ! Reaction with ammonia: $RCOCl + NH_3$ $\rightarrow$ $ RCONH_2+ HCl$ Product: The corresponding non N-substituted amide ! Reaction with an amine: $RCOCl + R^{'}NH_2$ $\rightarrow$ $ RCONHR^{'}+ HCl$ Product: The corresponding N-substituted amide !
- (2) The capture of the $Cl$ atom by an electronic gap reagent (eg $AlCl_3 $ ) leaves a $+ $charge on the $C$ atom
Then these substances can perform an electrophilic attack, for example on a benzene ring: Reaction with benzene in the presence of $AlCl_3$ (Friedel-Crafts reaction): $RCOCl + C_6H_6 + AlCl_3$ $\rightarrow$ $ C_6H_5COR+ H^+AlCl_4^-$ Product: The corresponding aromatic carbonyl derivative !
Name the end products obtained (by following the above steps) from the reactants: - Methanoyl chloride and isopropanol →
- Ethanoyl chloride and Methanamine →
- Methanoyl chloride and benzene (with $AlCl_3$) →