







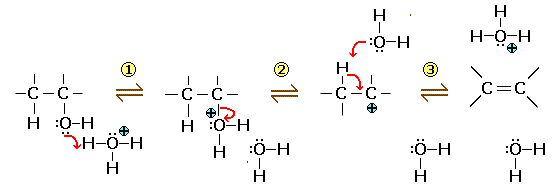
1) Attack of the hydronium ion on the $O$ atom of the alcohol and formation of an oxonium ion 2) Elimination of a water molecule and formation of a carbocation 3) α hydrogen capture by a water molecule and formation of an alkene, reformation of a hydronium ion

A carbocation is the more stable that it has more alkyl groups attached to it. This is because electron donating $ I+ $ effect partially fills the positive charge and makes it less active! This means that the ease of the dehydration reaction decreases from tertiary alcoholsto primary alcohols!