





Homogeneous catalysis is when the catalyst is in the same phase as the reactants and products.
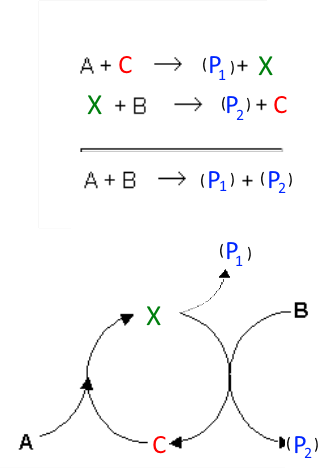
Often one of the reactants (here $A$) together with the catalyst (here $C$) produce optionally a product (here $P_1$ ) and an intermediate (here $ X $), which in turn reacts with the second reactant (here $B$) to optionally form a product (here $P_2$) and restitutes the catalyst. Example 1: Propagation step in the → radicalar substitution of alkanes: $Br\cdot$ $ + $ $CH_4$ $\rightarrow$ $ CH_3\cdot$ $ +$ $ HBr$ $CH_3\cdot$ $ +$ $Br_2$ $ \rightarrow$ $ CH_3Br$ $+$ $ Br\cdot $ with: $A$ $ =$ $ CH_4$ $C $ $=$ $ Br\cdot$ $B $ $=$ $ Br_2 $ $P_1$ $ =$ $ HBr$ $P_2 $ $=$ $ CH_3Br$ Example 2: Reaction of iodide and persulfate in the presence of some $Fe^{3+}$:
$S_2O_8^{2-}$ $+$ $2Fe^{2+}$ $\longrightarrow$ $ 2SO_4^{2-}$ $+$ $2Fe^{3+}$ $2Fe^{3+}$ $+$ $2I^-$ $\longrightarrow$ $2Fe^{2+}$ $+$ $I_2$ with: $A$ $ =$ $ S_2O_8^{2-}$ $C$ $ =$ $ Fe^{2+}$ $B $ $=$ $ I^- $ $P_1$ $ =$ $ SO_4^{2-}$ $P_2$ $ =$ $ I_2$
Many variants are possible, for example: In the case of → the reaction of hydrogen peroxide and potassium sodium tartrate things are getting more complicated:
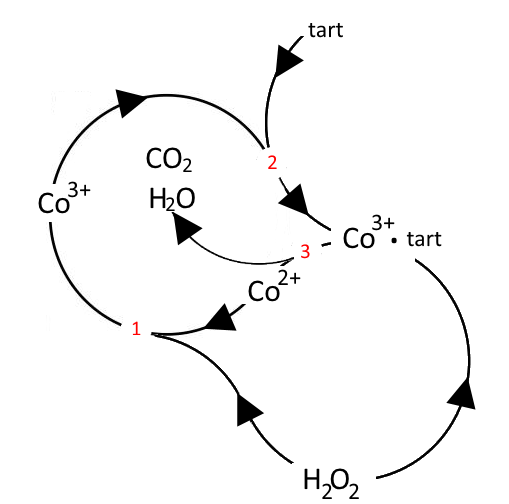
1 Oxidation of catalyst $Co^{2+}$ (rose) to $Co^{3+}$ 2 Formation of a the complex ion cobalt(III)tartrate (green) 3 Redox reaction: Tartrate and hydrogen peroxide are oxidised to $H_2O$ and $CO_2$ $Co^{3+}$ is reduced to $Co^{2+}$ (rose) and therefore restituted!
Detailed reactions are