





- Images are due to Georgia Perimeter College -
The electronegativity difference between $N$ and $H$ is low ($\Delta (E_n)$ $=$ $0,9$) compared for example to that between $O$ and $H$ ($\Delta (E_n)$ $=$ $1,4$).
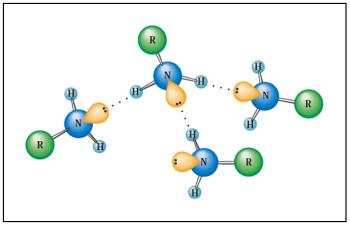
The weak hydrogen bonds between primary and secondary amine molecules are such that their volatility is less than that of alkanes, but greater than that of the corresponding alcohols:
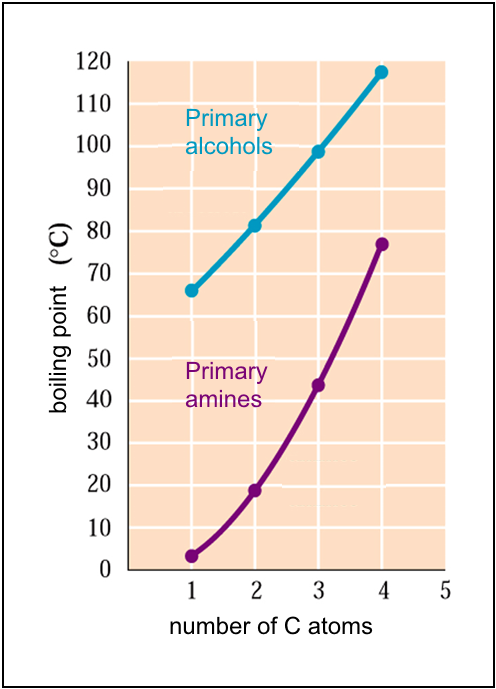 Thus, methylamine and ethylamine are gases at normal temperature, the methanol and ethanol not.
Thus, methylamine and ethylamine are gases at normal temperature, the methanol and ethanol not.
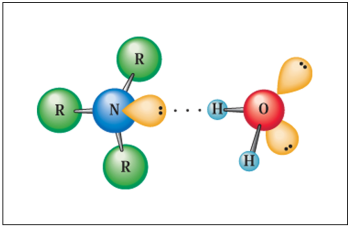
Primary and secondary amines can establish hydrogen bonds with water molecules, hence their solubility in water up to $C_5$ terms.