






Wallace Hume Carothers (1896 - 1937)
Wallace was the director of a research center of the firm duPont when he synthesized nylon in 1934.
This is a nucleophilic substitution reaction between an carboxylic acid dichloride (decanoyl dichloride or sebacoyl chloride) and a diamine (hexane-1,6-diamine or hexamethylenediamine) which leads to the polyamide nylon:
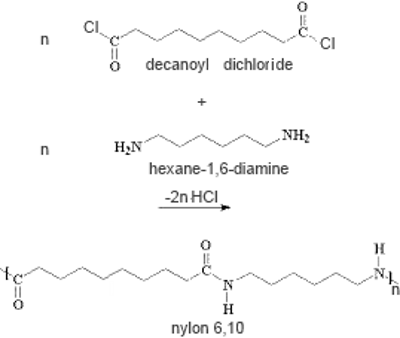
Adaptation of a video from KabiEdu
Remarks: - NaOH was added to the solution of 'amine' to neutralize the hydrochloric acid which is formed and which reacts with the amine to form the inactive ammonium salt . - The acid chloride is not added to the aqueous solution because it would react immediately to form the less active carboxylic acid. - Nylon formed at the interface prevents any further reaction, which is why it is removed for further synthesis.