





The pH of a dilute aqueous ethanol solution is 7. Ethanol has little acidic or basic character detectable by pH measurements. That's why you sometimes use ethanol as a solvent for pH indicators (eg phenolphthalein!)
In the Arrhenius theory of acids and bases, each chemical species that loses $H^+$ ion is an acid, that which earns one is a base:
- In the reaction with sodium, it is sometimes considered that alcohols lose $H^+$ of the hydroxyl group $OH$. This $H^+$ then serves to oxidize the sodium $Na$ to $Na^+$. The alcohol would be an acid:
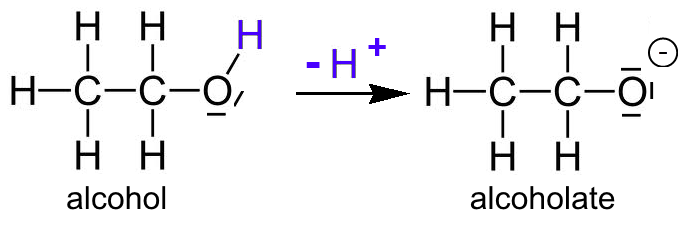 - In the reaction with sulfuric acid, $H^+$ from the acid is fixed on the oxygen of the alcohol hydroxyl group $OH$. The reaction generates heat. Alcohol would so be a base:
- In the reaction with sulfuric acid, $H^+$ from the acid is fixed on the oxygen of the alcohol hydroxyl group $OH$. The reaction generates heat. Alcohol would so be a base:
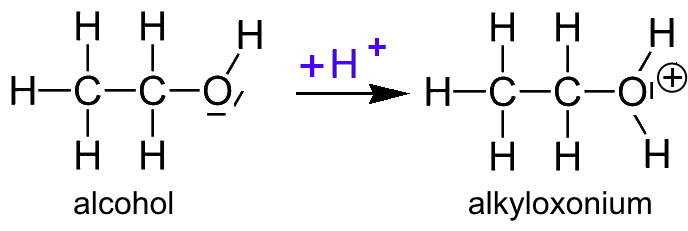
- The + I effect of the hydrocarbon chain increases the electron density between $O$ and $H$ and decreases the chances of $H^+$ to leave the alcohol. The more the chain is long and branched the greater the effect, the less will the alcohol be acidic.
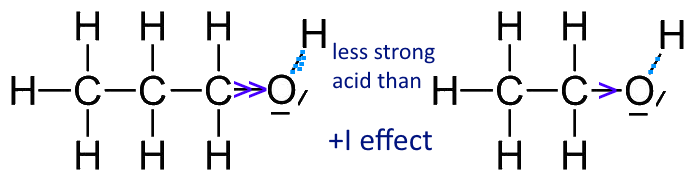 Propan-1-ol is less acidic than ethanol (provided one can speak of acidity in both cases)
- The alcoholate ions are the conjugated bases of alcohols. More they are stable, the greater will be their tendency to be formed and the more acidic the corresponding alcohol will be.
Propan-1-ol is less acidic than ethanol (provided one can speak of acidity in both cases)
- The alcoholate ions are the conjugated bases of alcohols. More they are stable, the greater will be their tendency to be formed and the more acidic the corresponding alcohol will be.
 Phenol has a marked acidic character because the phenolate ion is stabilized by mesomeric effect while cyclohexanol has no marked acidic character.
Phenol has a marked acidic character because the phenolate ion is stabilized by mesomeric effect while cyclohexanol has no marked acidic character.