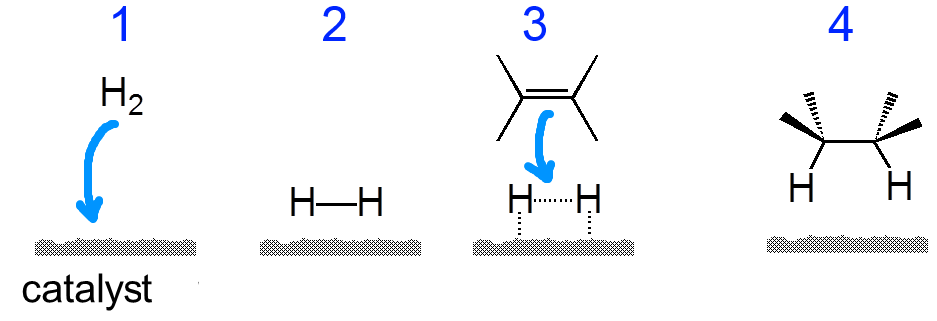
$H_2$ adds to an alkene in the presence of platinum or palladium catalysts or nickel/aluminum alloy.

The adsorption (1) of a molecule $H_2$ on an active site (2) of one of these catalysts activates molecule (3), allowing the addition (4):
$HCl,HBr,HI$ add easily on an alkene, $HF$ destroys the alkene

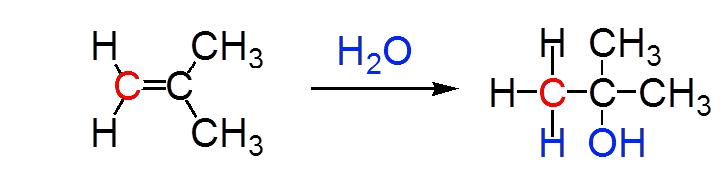
$H_2O$ adds at $150^oC$ in acidic medium

These additions are stereospecific:
Markovnikov's rule
 $H$ is always placed on the less substituted C atom of the C=C bond
$H$ is always placed on the less substituted C atom of the C=C bond
In red, the less substituted carbon (which carries least of substituents other than hydrogen):
1)
2)
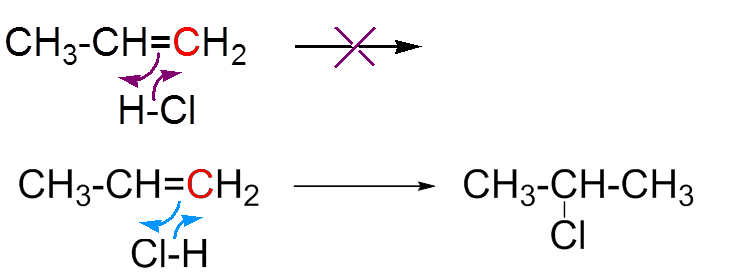
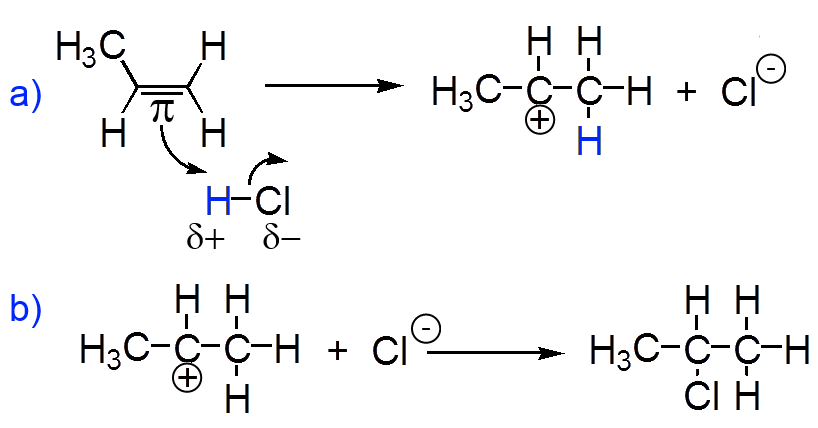
a) Electrophilic attack on the π doublet b) Capture of the chloride ion
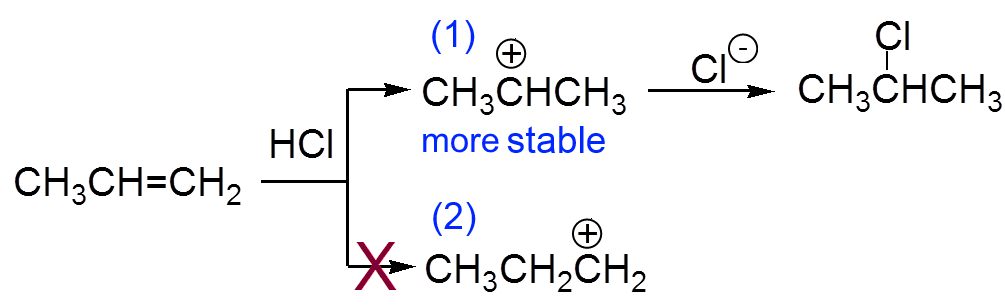
(1) is more stable than (2), because the inductive effect of the two methyl groups "fills" the positive hole better than the effect of ethyl. (1) therefore has a longer life, leaving the chloride ion time to get fixed.
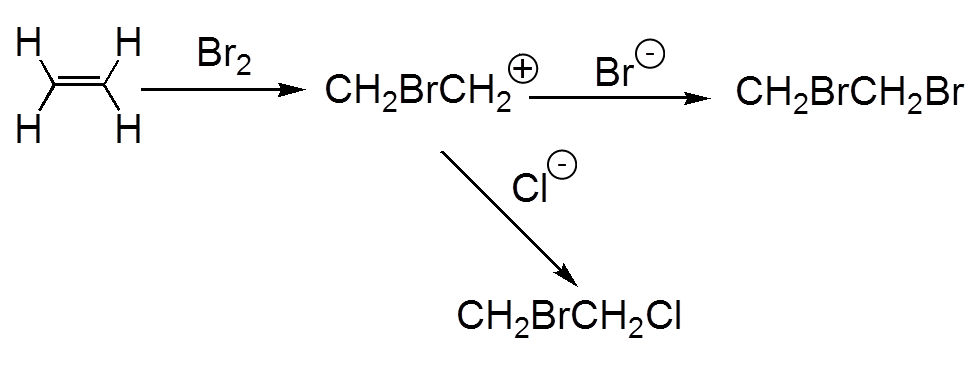
$Cl_2$ and $Br_2$ add easily on an alkene, $F_2$ destroys the alkene, $I_2$ is not added
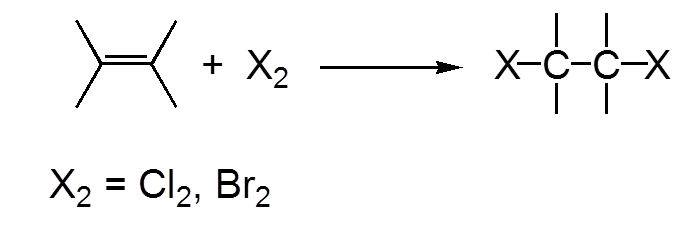
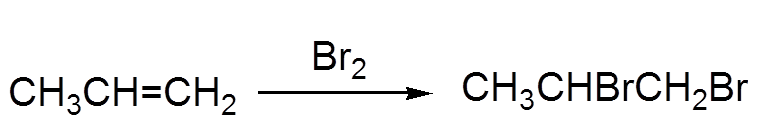
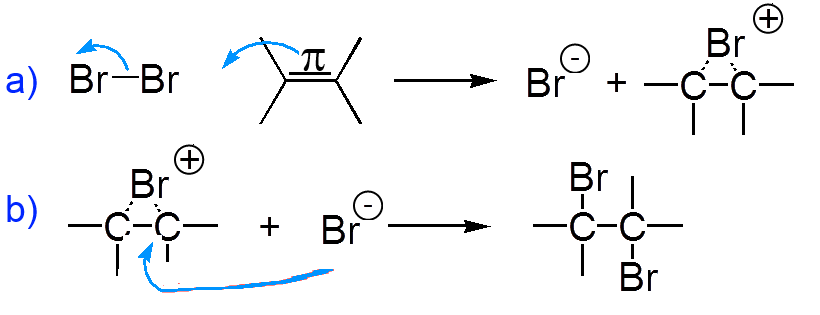
a) Electrophilic attack of bromine (electronegative) on the double π bond! b) Nucleophilic attack from "behind" of the bromide ion
There is formation of a bromonium ion intermediate. Note the multicenter bond that does not stick with the traditional Lewis structures.
The reaction of ethene on bromine performed in a solution of sodium chloride leads both to 1,2-dibromoethane and 1-bromo-2-chloroethane.