







The inductive effect is stronger the more the differences in electronegativity are stronger
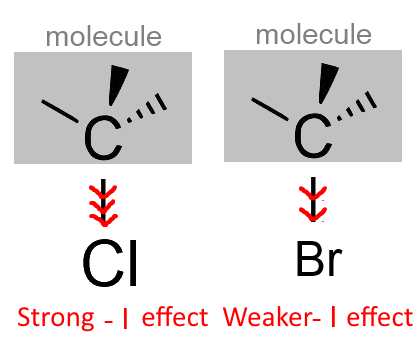
The greater electronegativity of bromine than chlorine causes a greater $-I $ effect.
$-I$ effect
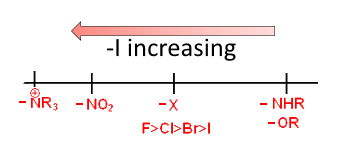
$R$ is an alkyl group or an $H$ atom. The more the hydrocarbon chain of $R$ is branched or long (in that order) the smaller is the $-I$ effect.
Strength decreasing from left to right: $OH $ $ \gt $ $ -OCH_3 $ $\gt $ $ $-OCH_2CH_3$ $ \ gt$ $ -OCH (CH_3)_2 $
$+I$ effect :
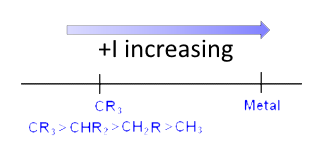
$R$ is an alkyl group or an $H$ atom. The more the hydrocarbon chain of $R$ is branched or long (in that order) the stronger is the $+ I$ effect.
Increasing strength from left to right: $-CH_3 $ $ \lt $ $-CH_2CH_3 $ $ \lt $ $-CH_2CH (CH_3) _2 $
 The effect on part of a molecule is even less strong the further the active group or atom (here $Cl$) is away.
The effect on part of a molecule is even less strong the further the active group or atom (here $Cl$) is away.
The inductive effect of a substituent causes (or modifies) a partial charge to where it is attached to the body of the molecule.

Here the partial charge on the $C$ atom decreases with the distance from the $Cl$ atom

Here the carbocation charge $+$ decreases with the size of the alkyl group.