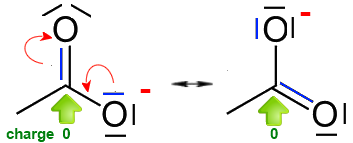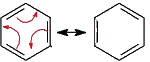
The arrows indicate where you need to move electron pairs from one contributing (resonance) structure to form another.
If one moves a σ doublet from a single bond, that would lead to a different molecule with a different arrangement of atoms!
Displacement a σ doublet is not allowed
As far as it does not lead to a forbidden structure, you can move own doublets only on neighbor single covalent σ bonds to form double π bonds.
Let's analyze the following movements:
 1
Displacement enabled
- on the next σ bond to form a π bond
- obtaining the perfectly allowed structure :
1
Displacement enabled
- on the next σ bond to form a π bond
- obtaining the perfectly allowed structure :
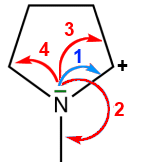
 2
Displacement forbidden
- On the next σ bond to form a π bond : OK
- The $N$ atom will have 8 electrons: OK
- The $C$ atom will have 10 outer electrons: not allowed
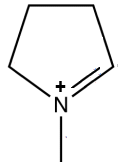
2
Displacement forbidden
- On the next σ bond to form a π bond : OK
- The $N$ atom will have 8 electrons: OK
- The $C$ atom will have 10 outer electrons: not allowed
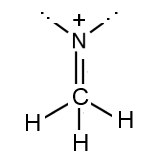 3
Displacement forbidden
- to a σ bond further away: not permitted
- The atom $C$ on top would have 10 electrons: not permitted
4
Displacement forbidden
- to another atom: not permitted
- This $C$ atom would have 10 electrons: not permitted
3
Displacement forbidden
- to a σ bond further away: not permitted
- The atom $C$ on top would have 10 electrons: not permitted
4
Displacement forbidden
- to another atom: not permitted
- This $C$ atom would have 10 electrons: not permitted
As far as it does not lead to a forbidden shape, you can move a π doublet - on a neighboring atom as own doublet - on the next σ bond to form a π bond
Let's analyze the following movements:
 1
Displacement permitted
- resulting in the following structure perfectly permitted (but not very contributory, see later):
1
Displacement permitted
- resulting in the following structure perfectly permitted (but not very contributory, see later):
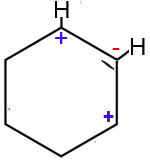 2
Displacement permitted
- on the next σ bond to form a π bond : OK
- The $C$ atom at the top will have 6 electrons: OK
- The $C$ atom at the bottom on the right side will have 8 electrons: OK
2
Displacement permitted
- on the next σ bond to form a π bond : OK
- The $C$ atom at the top will have 6 electrons: OK
- The $C$ atom at the bottom on the right side will have 8 electrons: OK
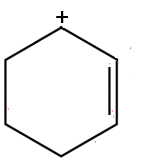 3
Displacement forbidden
- This is the movement of a σ bond which would change the structure of the molecule: not permitted
- Also the lower $C$ atom would have 10 electrons: forbidden
4
Displacement forbidden
- Displacement on a remote σ bond: not allowed
- In addition the two doubly bonded $C$ would have 10 electrons: forbidden
3
Displacement forbidden
- This is the movement of a σ bond which would change the structure of the molecule: not permitted
- Also the lower $C$ atom would have 10 electrons: forbidden
4
Displacement forbidden
- Displacement on a remote σ bond: not allowed
- In addition the two doubly bonded $C$ would have 10 electrons: forbidden
Rule: In all previous cases we find that
The atom on "the tail" of the arrow increases its charge by one, the one that is "at the forefront" reduces its charge by one .
Examples:
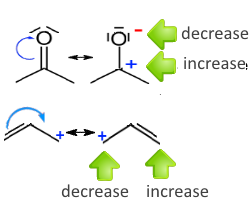
The displacement of several doublets is done according to the rules above, additionally:
If you move several doublets - they must be neighbors - the charge of the central atom does not vary - we must respect all previous rules
Example: