






Search:
By the mid-19th century, the two chemists Kekule and Ladenburg tried to determine the structure of a well-known molecule, benzene.

Benzene is a colorless liquid with a characteristic odor, irritating to skin and eyes, toxic and carcinogenic. Its molecular formula is $C_6H_6$
By substitution of an H atom of benzene with chlorine, we find only one single $C_6H_5Cl$. Kekule and Ladenburg have concluded that all $H$ atoms of benzene are "equivalent"
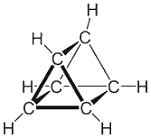
Ladenburg imagined benzene as a triangular prism where actually all $H$ atoms are equivalent:
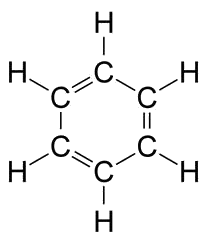
Kekule imagined (in a dream!) benzene as a hexagon with alternating single and double bonds where still all $H$ atoms are equivalent:
Here is an actual reaction of benzene that the structure of Ladenburg does not explain:
$C_6H_6$ $+$ $3H_2$ $\longrightarrow $ $C_6H_{12}$
Indeed, an alkane such as the single bonded "prismane" of Ladenburg does not add hydrogens, while the Kekule structure can explain the reaction:
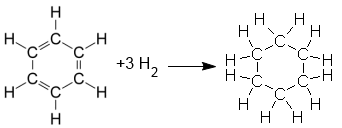
When Kekule was still alive, disturbing facts about its structure were presented that led half a century later to the notions of resonance or mesomerism .