





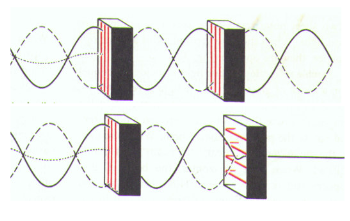
Two Nicol prisms are used to see if the plane of polarization of light determined by the first prism (polarizer) remained unchanged during the run up to the second (analyzer).

In the tank was introduced a solution of glucose, which we know is the one of two enantiomers with the same semi-structural formula. Here is the result:
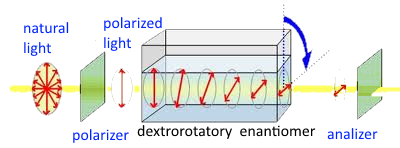
We see that the polarization plane was diverted to the right. Glucose is a natural dextrorotatory enantiomer . The other enantiomer (which we can synthesize) would be levorotatory
A wave train is determined by the 3 vectors: electric field, speed and magnetic field which form a dihedral angle:
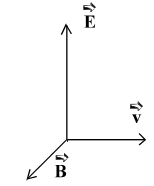
By exchanging two of these vectors, a chiral image is obtained! Two enantiomers can only be distinguished in the presence of a reagent or a chiral phenomenon itself, which in our case is the light
Example: Glucose
Dextrorotatory enantiomer: (+) - Glucose Levorotatory enantiomer: (-) - Glucose Formerly, we designated these two enantiomers by d - glucose and l - glucose, but we will see that this is confusing!