





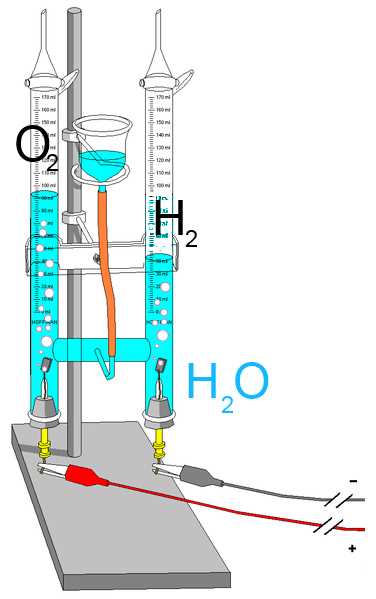
The Hoffmann apparatus is used as electrolysis cell. The two electrodes, cathode (-) and anode (+) immersed in acidified water are connected to opposite poles of a generator. - At the cathode hydrogen is produced - At the anode oxygen is produced $2H_2O$ $ \longrightarrow$ $ 2H_2$ $+$ $O_2$ The volume of the hydrogen is twice that of the oxygen.