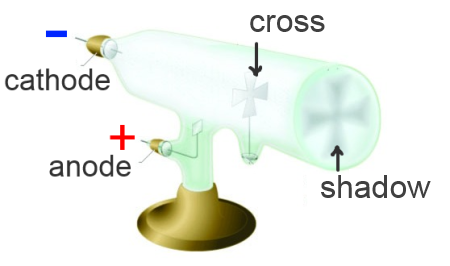
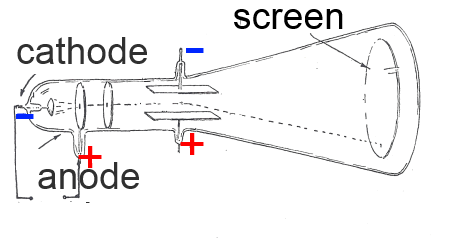
In the middle of the 19th century, Sir William Crookes had developed a tube which was very popular on the village fairs. Applying a high voltage to anode and cathode of the air-emptied tube, he produced cathode rays
:
Studying the deviation of cathode rays in an electric fied, Jean Perrin found that
Cathode rays are - particles with a very small mass - charged negatively - issued out of the cathode
Studying the deviation of cathode rays in an electric fied and in a magnetic field , Sir Joseph John Thomson proved and decided that
Cathode rays - are constituents of the atoms. - are called electrons.
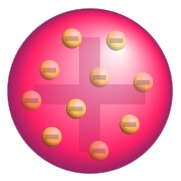
At that time Thomson assumed that atoms would be constituted of a positive matrix where the small negative electrons would be embedded like grapes in an english Plum Pudding: