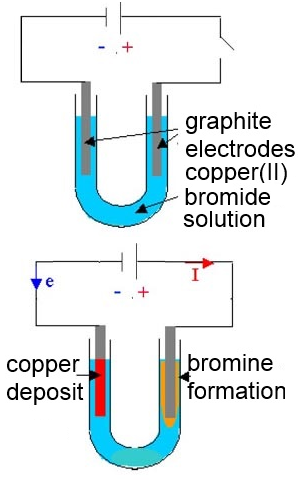
Electrolysis of a copper(II) bromide solution is done between (inert) graphite electrodes. Give the formulas of the ions which are in presence !
Ions: $Cu^{2+}$ ; $Br^{-}$ from dissolution and dissociation of copper(II) bromide $H^{+}$; $OH^{-}$ from the partial dissociation of water.
Look for the strongest oxidant - for the strongest reductant !
$Cu^{2+}$ is the strongest oxidant $Br^{-}$ is the strongest reductant.
Write the equations of the reactions occuring at the electrodes !
Cathode: $Cu^{2+}$ $+$ $2e^-$ $\longrightarrow$ $Cu$ (reduction) Anode: $2Br^{-}$ $-$ $2e^-$ $\longrightarrow$ $Br_2$ (oxidation)
What is observed?
At the cathode: deposit of metallic red copper on the electrode.
At the anode: formation of brownish-orange bromine which dissolves in water.