





When a partial positive charge δ+ is located on an hydrogen atom (small!) it produces a strong attraction between the molecules which is sometimes considered as a true "bond".
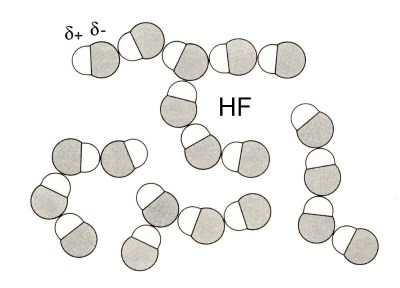 The attraction between hydrogen fluoride molecules is strong. Therefore one might consider to be in presence of much larger "pseudo molecules".
The hydrogen bonds are very important in biology and biochemistry because they determine the structure of → proteins or nucleic acids like → DNA
The attraction between hydrogen fluoride molecules is strong. Therefore one might consider to be in presence of much larger "pseudo molecules".
The hydrogen bonds are very important in biology and biochemistry because they determine the structure of → proteins or nucleic acids like → DNA
The boiling and fusion points of substances increase generally with the molar mass. Heavier molecules are indeed relatively more inert and therefore less inclined to move. Example: The non-polar alcanes:
| alcane | tB(oC) |
| CH4 | -161,7 |
| C2H6 | -88,6 |
| C3H8 | -42,1 |
| C4H10 | -0,5 |
Consider the following diagram:
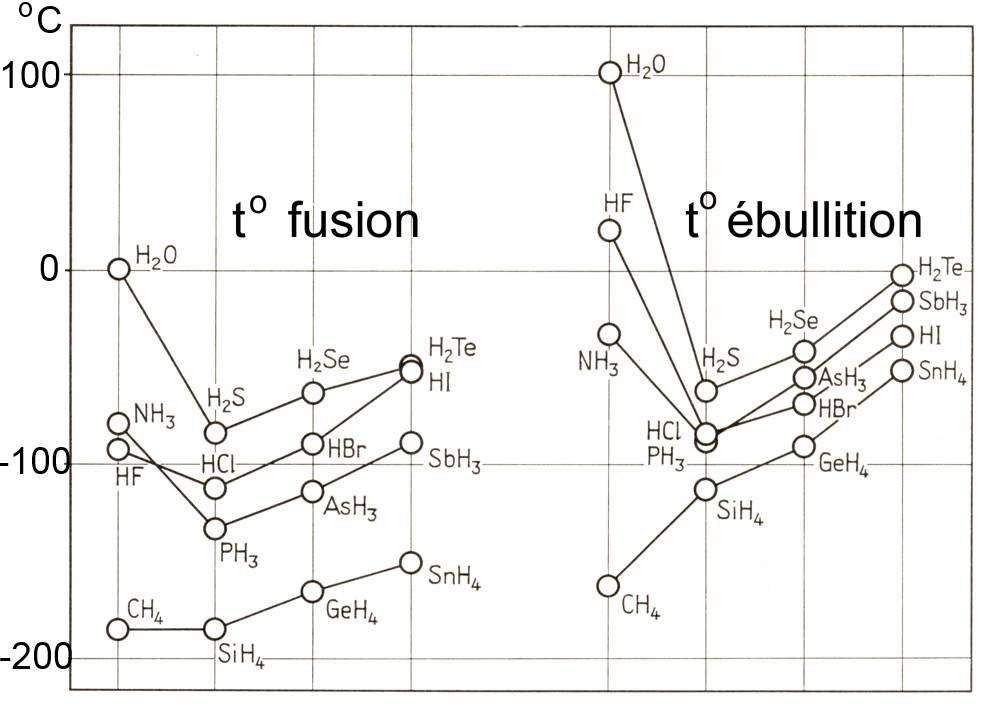 The diagram reveals that in general
The diagram reveals that in general
Due to polarity, polar substances have higher boiling and fusion points than non-polar substances of equal molar mass
- The boiling point of ammonia (polar, pyramidal structure!) is for instance higher than that of methane (non polar, tetrahedral symetrical structure!)
- Furthermore, the diagram shows that in the third period, boiling and fusion points increase with the molar masses too, for instance
toB(H2S)
< toB(H2Se)
< toB(H2Te)
- The polar substances of the second period present an anomaly, their fusion and boiling points are very high in spite of their small molar mass}, for example
toB(H2O)
>>toB(H2S)
in spite of a bigger molar mass of H2S !
The strong hydrogen bonds between those molecules are the cause of this anomaly!
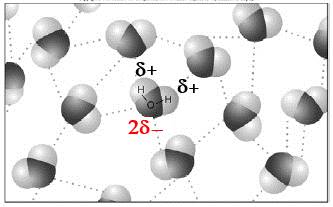 The polarity is therefore the cause explaining why water is liquid at the surface of the earth!
The polarity is therefore the cause explaining why water is liquid at the surface of the earth!
It is thanks to polarity that our earth is blue!
Most substances are denser in solid state than in liquid state.
That is not the case for water. In (water) ice, the hydrogen bonds force the H2O molecules in an hexagonal lattice with big gaps, which is therefore not so dense.
 So ice is less dense than water and swims on it.
If the contrary would be true, the oceans would always have frozen from the bottom to the top and life could never have developed there!
So ice is less dense than water and swims on it.
If the contrary would be true, the oceans would always have frozen from the bottom to the top and life could never have developed there!
Due to polarity, ice is less dense than water
Thanks to polarity, life has developed on earth!

Fuel (consisting of non-polar alcanes) and water (polar) don't mix (B) whereas polar substances like alcool and water(A) mix perfectly.
Polar and polar substances → miscible Non polar and non polar substances → miscible Polar and non polar substances → not miscible
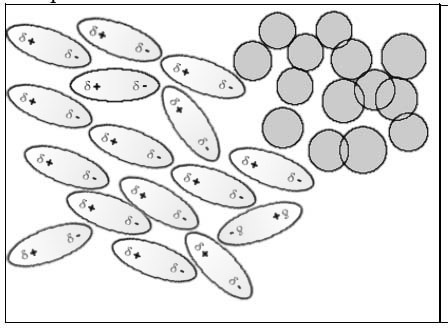
The mutual attraction of polar molecules prevents non-polar molecules to mix with them.
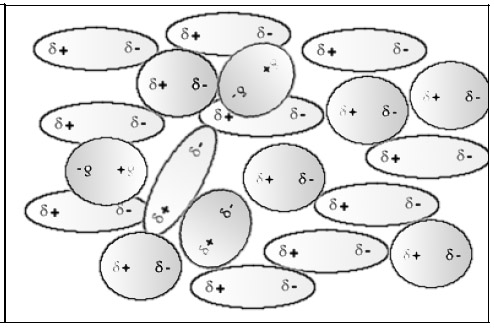 The molecules of both polar substances attract each other and interpenetrate.
The molecules of both polar substances attract each other and interpenetrate.
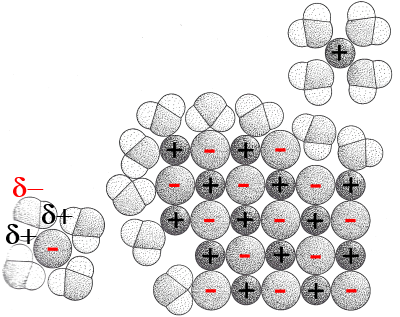
The image shows how a salt dissolves: The polar water molecules attach around positive ions (with their poles δ-) and negative (with their poles δ+). They say they hydrate ions. Hydrated ions can easily enter the water because of the polarity of the molecules H2O around them.
Without polarity, the soup would be fad!
If the ion cohesion in the ionic lattice is too strong, hydration followed by dissolution is impossible: substances are insoluble.
The metal cations often crystallize with their hydration water molecules staying attached to them:

The hydration of cations is a special case of a widespread phenomenon:
Small cations often surround themselves with polar molecules or anions to form "coordination complexes"
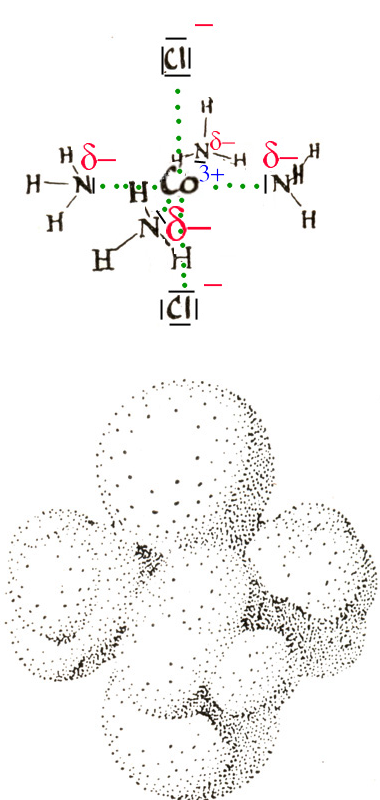
The central ion Co3+ is surrounded by 6 ligands : 4 molecules of ammonia (NH 3 of pyramid structure with δ - at the nitrogen atom) and two chloride ions. Many important molecules in biochemistry are coordination complexes, such as chlorophyll and hemoglobin.
Without polarity, no breathing, no green plants!
Many images of this page have been borrowed from the "chemistry bible" Linus Pauling: College Chemistry (Freeman, second edition)