





A covalent bond is polar if the difference of → electronegativities of the two atoms is different from zero

The chlorine atome is more electronegative than the hydrogen atom. It attracts rather on its side the two electrons of the common bond. The part of the HCl molecule around the chlorine atom presents a slight excess of negative charge (noted δ-), obviously smaller than a full negative charge, as in the Cl- ion. The part of the HCl molecule around the hydrogen atom presents a slight excess of positive charge (noted δ+), obviously smaller than a full positive charge, as in the H+ ion. We are in the presence of a dipole, a vector quantity directed from δ+ to δ- (dipole moment vector)
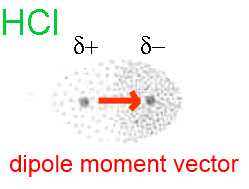
The intensity of the dipole moment vector depends on the difference of the electronegativities between the two atoms:
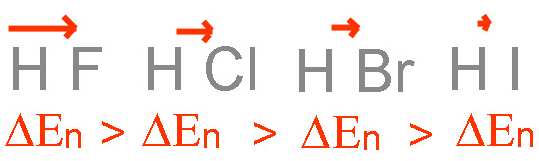
Molecules have often several dipole moment vectors. The addition of these vectors gives a resultant dipole moment vector.
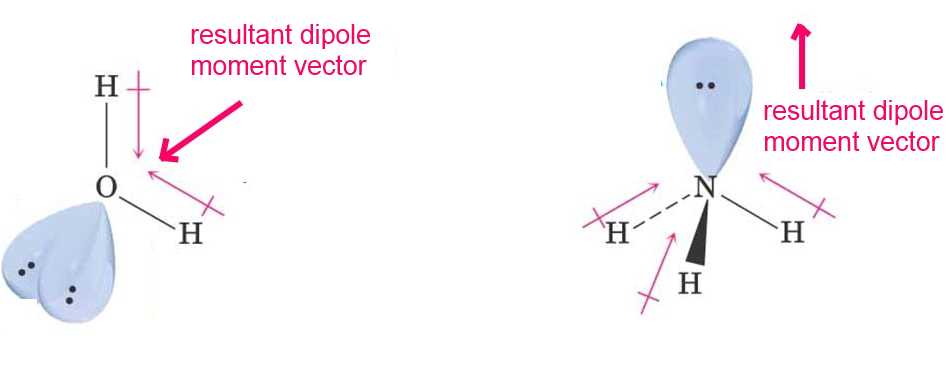
The water molecule has a resultant dipole moment vector directed towards the oxygen atom, the ammonia molecule has one directed to the nitrogen atom.
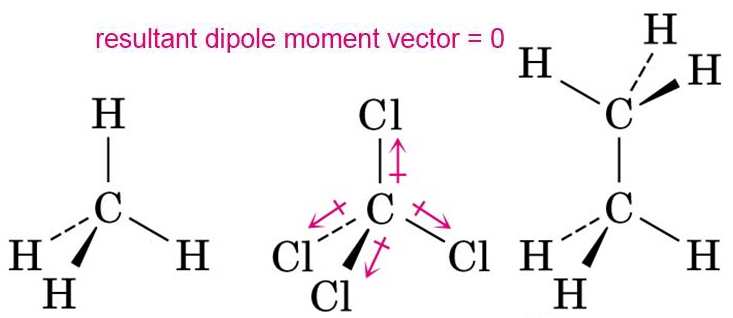 The dipole moment vectors of the molecules of methane, carbon tetrachloride and ethane result to zero because those molecules are in a symetrical arrangement.
The dipole moment vectors of the molecules of methane, carbon tetrachloride and ethane result to zero because those molecules are in a symetrical arrangement.
A polar molecule is a molecule with a resultant dipole moment vector different from zero A non-polar molecule is a molecule with a resultant dipole moment vector equal to zero
A polar substance is a substance whose molecules are polar A non-polar substance is a substance whose molecules are non-polar
Experimental evidence of a polar substance:
 The burette contains water, the ebonite stick has been charged negatively by rubbing.
The water shows to be attracted by the ebonite stick.
The burette contains water, the ebonite stick has been charged negatively by rubbing.
The water shows to be attracted by the ebonite stick.
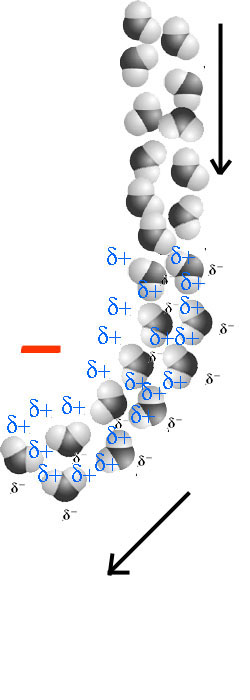 Influenced by the negative charge of the ebonite stick, the water molecules toggle while falling down, their δ+ located on the H atoms towards the negative charge.
Since the distance of the δ+ to the negative charge is smaller than than the distance of the δ-, the attraction force is bigger than the repulsion force, therefore the water molecules are attracted towards the ebonite.
Influenced by the negative charge of the ebonite stick, the water molecules toggle while falling down, their δ+ located on the H atoms towards the negative charge.
Since the distance of the δ+ to the negative charge is smaller than than the distance of the δ-, the attraction force is bigger than the repulsion force, therefore the water molecules are attracted towards the ebonite.
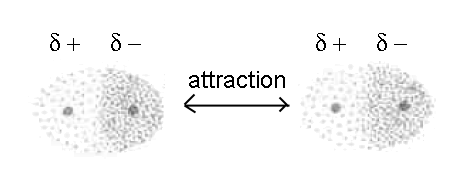
The attraction is the more strong the more the partial charges δ+ et δ- are big and the distance between the atoms is small.