





- Solid state (ice or snow), - Liquid state (liquid water) and finally - Gaseous state (water vapour) are the physical states of the substance water. Physical state changes are :
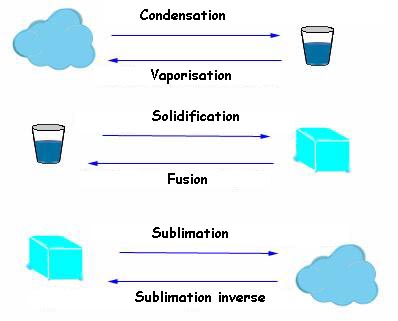
The same water molecules have quite different distribution in their three states:
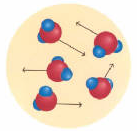
Gas: Molecules are distant from each another. Movement is quick and chaotic. Shocks
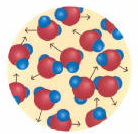
Liquid: Molecules are in contact. Movement is slow and chaotic. Mutual exchanges
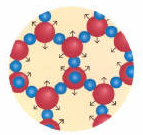
Solid: Molecules are in contact. Ordered state. Vibrations
Most liquids contract while temperature drops. Water is different. Water contracts until 4 oC, then it dilatates until solidification. Ice is less dense than liquid water, because the ordered state of ice molecules implies gaps which are filled in the disordered state of water molecules. If water behaved like other liquids, then there would exist no iceberg, ponds would freeze from the bottom to the top! On earth we find water in its three states. Earth is therefore a very special planet where temperature and pressure are just fitting exactly to permit these states.
The water molecules are attracting themselves mutually. That is called cohesion. Other materials may also attract water molecules. That is called adhesion.

Surface tension is cohesion of molecules of water in surface. Place a drop of water on wax paper. Examine the drop. Why does it have this shape? What is going on? Water molecules are not attracted by wax paper. Every water molecule is attracted by other molecules of the droplet. That explains why water minimizes its volume and tends to adopt a sperical shape. It is as if molecules on the surface acted like a "skin" retaining the other molecules: Surface tension.

Place gently a clip on a water surface. It is floating. What is happening? The following images show the effect of surface tension:
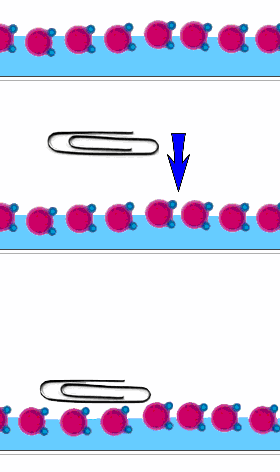
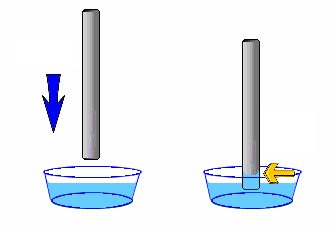
Surface tension is due to the cohesive property of water. Capillarity by contrast, is due to the adhesive property. Place a transparent straw in a water glass. Examine the level of the water in the straw: Water climbs up in the straw!